Special Report
Neuroscience of need
Understanding the addicted mind
By Bruce Goldman
Photographs by Kelly Hennigan
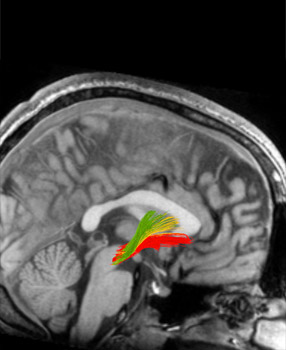
In this MRI of a brain (side view), the green, yellow and red areas indicate bundles of neurons involved in addiction. Red represents reward pathways; green and yellow signify habitual responses.
In the past 10 or 15 years, there’s been a shift in thinking about addiction, to a new appreciation that it is, at its root, a maladaptive form of learning. And like learning to ride a bike, addiction is not quickly unlearned.
If you think quitting is a simple matter of willpower, you’re in good company. More than a third of the general public agrees, according to a 2008 survey by the federal Substance Abuse and Mental Health Services Administration. But it’s tougher than that.
“It’s kind of like putting on a lot of weight,” says Keith Humphreys, PhD, a Stanford professor of psychiatry and behavioral sciences who has served as a senior White House drug-policy advisor. “Your body changes, and from then on losing weight is way harder than it ever was before you got fat in the first place. Because addiction-associated brain changes are so enduring, a lot of people are going to relapse. So the course of treatment has got to be longer-term than it often is.”
Some of the key biological insights were made by Stanford neuroscientist Rob Malenka, MD, PhD, who continues his studies using animal models to extrapolate to humans. And now others, like brain imaging expert Sam McClure, PhD, are finding that changes Malenka sees in rats take place in humans as well.
This new understanding of addiction’s long-term grip has policy implications: A short-term detox stint to rid the body of the unwanted chemical just won’t cut it. Authorities have to be prepared to treat addiction as they would any chronic disease, even though that implies long-haul and therefore costlier treatment (it’s still a lot cheaper than imprisonment). An equally important implication: They must also try their best — from both health and cost standpoints — to prevent people from starting down that lonely, dangerous road in the first place.
Unforgettable
There are things you don’t forget, and there are things you can’t. For people who become drug addicts, the drug experience — the substance, but the entire “scene” too — is not only unforgettable but indelibly etched into the physiological brain circuitry that drives us onward through the obstacle course of existence.
And much of that memory is false. Because all addictive drugs appear to share a rather mysterious property: They’re “better than the real thing.” Better, that is, than the real things our reward circuitry was designed by evolution to reward: food, sleep, sex, friendship, novelty, etc. And better, even, than they were the last time around. At least, it sure seems that way to the addict.
About 25 million Americans are addicted to drugs (including alcohol but excluding nicotine), about the same number as those who have diabetes. But wanting a drug — really, seriously craving it — doesn’t mean you have to like it. “That’s a big part of the problem of addiction,” says Malenka, the Nancy Friend Pritzker Professor in the Department of Psychiatry and Behavioral Sciences. Malenka was among the first investigators to home in on the molecular details of just how the mechanisms involved in memory and learning are hijacked by drugs of abuse.
Addictive drugs mimic natural rewards such as food and sex by kindling a network of brain areas collectively called the reward circuitry, which is responsible for enjoyment — which if you think about it, is an important survival response. It gets us to do more of the kinds of things that keep us alive and lead to our having more offspring: food-seeking and ingestion, hunting and hoarding, selecting a mate and actually mating.
Moreover, addictive drugs fire up the reward circuitry in a way that natural rewards can’t — by, in a sense, pressing a heavy thumb down on the scale of pleasure. Over time, the desire for the drug becomes more important than the pleasure the addict gets from it. By the time the thrill is gone, long-lasting changes may have occurred within key regions of the brain.
The brain is a little bit like the big snarl of tangled wires snaking their way out of that six-outlet surge protector behind your bed. They know where they’re going, even if you don’t. Nerve cells (or neurons, as scientists call them) can be seen as hollow wires transmitting electrical currents down long cables called axons to other neurons.
Addiction was once defined in terms of physical symptoms of withdrawal, such as nausea and cramps in the case of heroin or delirium tremens in the case of alcohol, which reflect physiological changes within cells of an addict’s body. It’s now seen as changes in brain circuits, or combinations of neurons; in other words, the very neurophysiological changes that result from learning and experience. You crave, seek and use a pernicious drug again and again because you have a memory of it being more wonderful than anything else, and because your brain has been rewired so that, when exposed to anything that reminds you of the drug, you will feel rotten if you don’t get some.
“These are symptoms of a brain disease, not a mere weakness of will,” Malenka says. He and other researchers are working to understand addiction as a sum of behavioral consequences of changes within nerve cells that occur with repeated drug use. Over time, these subcellular changes alter the strength of connections in the circuit, essentially hardwiring the yen for drugs into a habitual craving that is easily reignited not only by the drugs but also by environmental cues — people, places, things and situations associated with past drug use — even when the addict hasn’t been anywhere near the drug or the drug scene for months or years.
Serendipity strikes
In the 1950s James Olds, PhD, a postdoctoral researcher working with psychologist Peter Milner, PhD, at McGill University in Montreal, was conducting experiments to try to assemble a wiring diagram for some of this complicated brain circuitry. They were using a then-new technique, based on the understanding that neurons are at heart electrical critters, that came down to sticking electrodes (painlessly) into a rat’s brain, running an electric current and seeing what happened.
Your brain has been rewired so that, when exposed to anything that reminds you of the drug, you will feel rotten if you don’t get some.
At one point Olds and Milner were shooting for an area of the brain called the reticular formation, an archipelago of interconnected clusters dispersed throughout the brain and involved in arousal and attention. But they missed and hit another circuit by accident. They discovered that when they stimulated this circuit, the animals loved it.
So the investigators tried something new. They taught the rats to press a lever in order to deliver shocks to their own brains, and recorded the points in the brain that rats liked to electrically stimulate over and over again by pressing that lever — and press it they would, sometimes for hours on end, to the exclusion of just about anything including eating or drinking. (Of course, the rats couldn’t move the electrodes from one part of their brain to another. So Olds and Milner did that for them.)
Point by point, Olds and Milner were able to map the network of brain regions, interconnected as they are by bundles of axons running from one region to the next, that became known as the reward circuit. To oversimplify things a great deal, this circuit includes nerve bundles that run from deep inside the brain to spots such as the nucleus accumbens (associated with pleasure), the more recently evolved prefrontal cortex (involved in decision-making, planning and so forth), and other places of more ancient evolutionary vintage that control habitual movements and are sometimes referred to as the “lizard brain.”
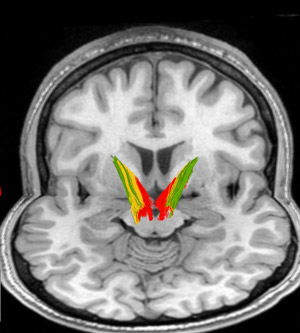
A brain, viewed from behind in two planes, showing the pathways implicated in addiction. The pathway travels from dopamine neurons (central) to areas in the striatum.
But what flips on the reward circuit in regular life, when electrical zaps to the brain are blessedly few and far between? The same chemical that’s triggered by dope. It’s called dopamine.
Dope fires up your dopamine
Dopamine is one of a growing number of known neurotransmitters, substances neurons produce for the purpose of relaying information from one neuron to the next. Different groups of neurons manufacture different neurotransmitters, which all work pretty much the same way but in different nerve bundles and with a spectrum of different results. These substances are stored inside numerous tiny bulbs budding from points along a neuron’s long, electricity-conducting axon at key contact points the neuron shares with other neurons.
When an electrical signal roaring down the axon’s surface rumbles past one of these little bulbs, myriad molecules of neurotransmitters get squirted into the surrounding space. They diffuse across that space (called a synapse) to specialized receptors on the abutting neuron, where the interaction can either set off (enhance) or shut down (impede) a new electrical current in the downstream neuron.
These dopamine-squirting neurons constitute a tiny fraction of all neurons. But each of them can network with up to 10,000 or more other neurons stretching to the far corners of the brain. A dollop of dopamine in your tank can really boost your reward mileage, so to speak.
Once dopamine’s centrality to the neurons constituting the reward circuit was worked out, people started wondering whether drugs might activate the reward circuits. It turned out that they do.
“One reason that the advances in our study of the neurophysiology of addiction so far exceed our understanding of other psychiatric disorders is because the animal models for addiction are extremely good,” says Malenka. Teach a rat to press a lever for an infusion of a drug of abuse, and you will see the same compulsive behavior in the rat that you would in a person. “A rat will work very hard to get drugs,” he says. “It will press that lever hundreds, even thousands, of times and endure pain and suffering to get drugs.”
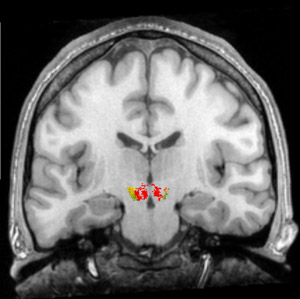
In this rear view of the brain, the colored areas show the origin of the dopamine neurons in the midbrain.
As these animal studies have shown, virtually all abused drugs — for instance, heroin and other opiates; cocaine, amphetamines and other psychostimulants; nicotine; and alcohol — operate by interfering with the reward circuitry. They cause the release of dopamine in target structures such as the nucleus accumbens, that key structure in the experience of pleasure.
Different drugs do this in different ways. Cocaine and amphetamines prolong the effect of dopamine on its target neurons. Heroin inhibits other neurons that inhibit these dopamine neurons. (In the logic circuitry that is the brain, a double negative roughly equals a positive.)
Hijacking the reward system
You might think that the more you eat, or the more sex you have, or the more good vibrations you get, the more dopamine your reward-circuit neurons will squirt at their target structures in the brain. But it’s not so simple.
A seminal 1997 Science paper by P. Read Montague, PhD, at Baylor postulated that what really gets the reward circuitry jazzed up isn’t so much the good vibes as it is the extent to which the goodness of the vibes exceed expectations.
The newer theory was based on animal studies involving lever pressing, with a twist. In this case, the test animal learns that if it presses a lever after it receives an environmental cue — to wit, a light goes on — it will get a reward: say, a nice slice of apple or a drop of juice, both of which rats love. Of course, the animal soon learns to reach for the lever the instant the light goes on. With repeated exposure, the rat gets the hang of it, and a few interesting things happen inside its brain. First of all, the reward itself (the food) no longer produces the dopamine surge associated with reward-circuit activation.
Second of all, it is now the light, not the food, that triggers the activity in the reward circuit. The timing of the reward-circuitry’s dopamine squirts has shifted from the time of reward delivery to the time of the cue (the essence of the so-called “conditioned response” familiar to anyone who has ever taken Psychology 101).
It’s not that the juice or apple slice no longer tastes good. It’s that the reward circuitry is responding to the difference between what we expect and what we get. How much dopamine gets secreted depends not on how great the reward is, but on the degree to which it meets expectations. The juice still tastes great, but it’s no longer a surprise; it’s predictable. However, the light’s timing can’t be predicted. It’s always a surprise, and (as the animal now knows) it’s always a prelude to something good.
The reward circuitry is always secreting dribs and drabs of dopamine. If an experimental animal gets a bigger-than-expected reward, the frequency and amount of dopamine secretion increases; if it’s smaller than anticipated (or if the light goes on but the animal’s frantic lever-pressing brings no juice at all), dopamine secretion drops below baseline levels. Moreover, this depression in firing rates of dopamine-secreting neurons occurs precisely when the anticipated reward should have come, but didn’t. Thus, the brain seems to interpret the absence of the expected reward not merely as a lack of enjoyment but as a punishment. (How does a rat spell “disappointed”?)
Sam McClure, an assistant professor of psychology at Stanford who studied under Montague, has been imaging human brains to visualize connections between the regions that constitute the reward circuit. “Variations in dopamine levels tell all kinds of structures in your brain when something you want is within reach, getting closer, slipping away or not working for you anymore,” he says.
At least that’s the way it’s supposed to work. Cocaine, heroin and other abused substances usurp this system. And they do it in a really creepy, pernicious way: by short-circuiting it.
With normally rewarding things like food and sex, we usually have a pretty good idea of how good it will be. It’s when the reward exceeds our expectations that the dopamine circuitry really lights up big time. Conversely, if our expectations aren’t met, dopamine activity drops off.
But cocaine, heroin, alcohol and nicotine directly activate the circuit — they goose dopamine secretion — regardless of how high the expectation was. “Every time you take it, you activate that dopamine activity, so you’re getting a readout that says, ‘Wow, this was even better than I thought it would be,’” McClure says. “It’s always better than you expected. Every single time.” The experience is remembered as always getting better — even if, paradoxically, it’s actually not so great anymore. (“Tolerance mechanisms” within the brain can cause a drug’s pleasant effect to diminish with repeated use.)
The needle and the damage done
In susceptible individuals, repeated drug use creates the same kind of lasting changes in the connections among neurons that we get from learning to ride a bike.
One important way our brains snap an experience into long-term memory is by strengthening the synaptic contacts between neurons in the network that encodes this experience. This involves a number of biochemical changes in both the bulb protruding from a neuron’s axon and the brush-like extension of a nearby neuron. Drug abuse can also cause neurons to sprout brand-new synapses — for example in the nucleus accumbens, the hotspot for positive emotions. It can weaken synapses, too. Nora Volkow, MD, of the National Institute on Drug Abuse has shown that the plan-oriented prefrontal cortex functions poorly in cocaine addicts.
The long-term strengthening of drug-associated memory circuits, combined with that “even better than expected” illusion addictive drugs foist on users, goes a long way toward explaining what is probably the biggest problem addicts and those who treat them face: a pronounced tendency to slide back into the habit. It’s why former White House drug-policy advisor Humphreys believes long-term treatment is vital.
If you are an addict, not just the drug but also all the associated physical, geographical and social cues exert a powerful effect, even decades after the last time you were anywhere near the drug: You walk past the bar you used to get drunk in and see your buddies in there, or you smell cigarette smoke — or, if you used to inject cocaine or heroin, all it may take is seeing a spoon — and you experience a craving and risk a relapse. Stress — you lose your job, suffer a divorce, undergo a financial crisis — can mimic drugs’ influence on the reward circuitry, and is therefore another major cause of relapse.
Who’s susceptible? (Who knows?)
Only a fraction of people who experiment with drug use get addicted. But virtually all of us have an intact, functional reward system. So why wouldn’t we all be subject to the tyranny of drug-induced illusions of “better-than-expected-ness?”
The short answer is that nobody knows enough to be able to single out a potential addict with any certainty. “There’s no such thing as an ‘addictive personality,’” says Humphreys. “Those 25 million addicts in the United States have wildly different personalities.” There are, however, obvious risk factors: genetics, poor social support networks, a sense of having nothing to lose and stress.
One big risk factor, says Humphreys, is the age at which you start using. “We’re the most vulnerable to addiction in our early teenage years, when our brains are most plastic. So it’s not an accident that almost every single adult smoker started smoking when they were teenagers. If you start smoking when you’re 30, you are almost certainly not going to get addicted. But the younger you start, the more likely you are to keep smoking.
“There are two groups of people who really understand that: prevention professionals, and the tobacco companies. You want to make addictive substances as inaccessible as possible in the environment, particularly for young people.”
The biggest risk factor of all, of course, is the initial use of an addictive substance, says Malenka. “It’s impossible to get addicted if you never take the drug.”


