SUMMER 2008 CONTENTS
Home
Good as Gold?
New drug approvals ebb; doubts over testing's gold standard grow
Q&A with Katie Couric
Standing up for cancer research
Breath of Hope
Lifeline or gamble? Sometimes a clinical trial is both
Just Another Lab Rat
The human subjects trade is booming, largely without oversight
Fixing Trial Tribulations
Solutions from Stanford
A Spoonful of Sugar Pills
Why nothing really is something, and in some ways is better than anything
Banding Together
Minds of all kinds join to hasten discoveries of new medical treatments


Cuff 'em
Locking tumors in their boxes
By Elizabeth Crown
Photo by Duncan Stewart
Science writers anthropomorphize the behavior of cancer as it spreads, likening malignant cells to marauding barbarians who run amok, colonizing and ultimately destroying the body.
To an extent, this description is apt. Cancerous tumor cells flaunt growth controls and invade normal tissues. Some tumor cells proliferate wildly by hijacking genes and enzymes involved in wound healing. Others roust genes that normally retire after embryonic development. After subverting those genes, cancer cells multiply and establish beachheads in foreign, potentially harsh locales.
Janine Erler
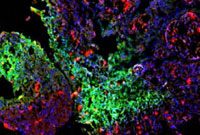
Inside a lung tumor (magnification x100): Low-oxygen regions are green, cell nuclei are blue and Lox, a molecule that aids tumor spread, is red.
But as it turns out, metastasis, which is responsible for 90 percent of deaths from solid tumors, is not chaotic. An emerging body of research from the laboratory of Amato Giaccia, PhD, shows that malignant tumors collude with their environment to dispatch cancer cells far and wide.
And now he’s fingered one protein he’s convinced is the Achilles heel of metastasis. Giaccia’s excited about the implications of his research for people with cancer. “We envision that, instead of being a fatal illness, cancer will become a chronic condition like diabetes or arthritis,” says Giaccia, professor of radiation oncology.
The metastatic tumor simplified
Giaccia notes that some of the greatest successes in cancer therapy have been in the treatment of blood malignancies — cancers without tumors — such as leukemias, lymphomas and Hodgkin’s disease. Researchers can also offer effective treatments for certain germ cell cancers, such as testicular cancer. But few cures exist for the majority of solid tumors.
So, what’s the world of a typical tumor? The mass of cancer cells sits surrounded by protein filaments and inflammatory immune cells, and abnormal — tangled and highly permeable — blood vessels, imbedded in a structure called the extracellular matrix.
“Like a Jell-O mold with fruit cocktail in it. The Jell-O is the matrix and the fruit is the tumor,” Giaccia says.
Metastatic tumor cells are tough. They must overcome physical barriers, including the matrix and the stalwart “basement membranes” that sheath many organs.
They also face physiologic barriers, such as limited oxygen and nutrients, higher acidity, and impaired access to the blood supply — all conditions that hinder normal cells. And then the immune system straight out attacks the tumor cells. Yet tumor cells thrive and spread.
Tumor cells hold an impressive bag of tricks. For example, they let loose enzymes that metabolize normal cells and dissolve the material between them, which allows them to spread into new tissues.
And they order other cells around. Some help comes from blood vessels, which create a supportive nest. Immune system cells, especially macrophages, carry messages from the tumor to the surrounding cells and promote reconfiguration of the matrix. Macrophages are also part of the cellular road crew that constructs the “metastatic highway” a tumor uses to migrate to new tissues.
But perhaps the tumor’s biggest trick is thriving in environments bereft of oxygen.
The lowdown on oxygen
It was in 1955 that scientists first realized solid tumors and their immediate surroundings have extremely low oxygen levels — or in the language of medicine, their cells and environment are hypoxic. This group of British investigators, led by Louis Harold Gray, PhD, at Mount Vernon Hospital, in England, also showed that hypoxic tumors resist radiation therapy.
In the decades that followed, researchers learned more about hypoxia’s impact. Among the highlights: The most aggressive cancers originate from hypoxic sites, and hypoxia decreases the effectiveness of chemotherapy and even surgery.
By 1996, Giaccia was wondering whether hypoxia imposed a strong selective pressure on tumor cells to adapt to their austere surroundings. Research published in 1990 by Richard Hill, PhD, senior scientist at the Ontario Cancer Institute, had hinted at this by showing that tumor hypoxia increased the production of genes that control metastasis.
So the Giaccia lab pursued this by zeroing in on a particular gene: a tumor suppressor known as p53. Are tumor cells that carry mutated p53 genes better suited to survive and adapt to a low-oxygen environment? The answer is yes, based on the experiments of Tom Graeber, PhD, who was then a graduate student in the Giaccia lab (he is now a UCLA postdoctoral fellow). Hypoxic conditions gave an edge to cells with less-than-ideal abilities to suppress tumors.
“Those two papers — Hill’s and ours — set the stage. Not only was hypoxia bad for therapy, it could affect malignant progression and metastasis,” Giaccia says.
Further scrutiny of how hypoxia affects gene activity in tumor cells led to a finding that could be cancer’s undoing. In 2003, Nicholas Denko, PhD, then a postdoctoral scholar in the Giaccia lab (and now an assistant professor of radiation oncology at Stanford), conducted a gene microarray experiment to see how genes alter their expression in response to oxygen. The result revealed that in tumors, hundreds of genes change their activity when the oxygen is low.
Finding a target
It was January 2004 and Janine Erler, PhD, had just begun her work in the Giaccia lab. Like every new postdoc, she received a list of the hypoxia-regulated genes, and a command to pick the one that interested her and explain why.
She picked lysyl oxidase, aka LOX, because of new research showing the molecule was present in aggressively metastatic cell lines.
“It made sense to me that if oxygen-deprived cancer cells were more invasive and metastatic, and they also have high levels of LOX, it may be that LOX is responsible for the increased invasive capacity,” says Erler.
Since then, Giaccia, Erler and colleagues have shown that inhibiting the LOX protein significantly impedes and sometimes even eradicates malignant growth in animal cancer cells and in human cancer cells transplanted into mice — and caused no negative side effects. In cases where metastasis already exists, inhibiting LOX kept cancer spread in check. And a look at the other side of the coin revealed that cancer patients with heightened levels of LOX had decreased survival.
As findings about LOX mount, its role in a tumor’s spread becomes clearer. Normally LOX helps form and stabilize the extracellular matrix, which, in addition to acting as a scaffold, regulates how cells communicate with each other. But in hypoxic conditions, a tumor cell’s production of LOX increases, which directs cells to build tracks for tumor cells to move through the body, and to prepare hospitable nooks and crannies for new tumors.
Not surprisingly, researchers outside Stanford are eyeing the results with interest. Giovanni Melillo, MD, an expert on tumor hypoxia at the National Cancer Institute, calls the laboratory work elegant and adds: “More important, the Giaccia group has demonstrated that the LOX pathway is relevant to human cancers and that it can be exploited for therapeutic purposes.”
Giaccia believes that anti-cancer therapies of the future will come not from targeting cancer cells, but by undermining their elaborate cellular support network. In other words, by targeting proteins like LOX — a strategy he hopes to see tested in patients if further animal studies raise no red flags.
He suggests that anti-LOX therapy could be combined with chemotherapy and/or radiotherapy — an antimetastatic “cocktail” — to target both a primary tumor and metastasis. His group is currently developing hypoxia-targeted substances that selectively amplify the effects of radiation and chemotherapy on tumor cells while sparing normal cells.
And measurement of LOX levels in the blood might eventually be used in the clinic as a prognostic and diagnostic marker of metastasis — a new area of molecular medicine called “theranostics” in which the disease marker is also the therapeutic target, and the activity of drug targets in the patient’s tissue or blood can be measured.
The end of metastasis?
Though most efforts in cancer research focus on eradicating primary tumors, the majority of deaths from cancer result from metastasis. And in some cancers, such as pancreatic, metastasis is usually under way by the time a primary tumor is first detected.
But new research findings indicate that strategies targeting hypoxia-related proteins could turn this around, says Giaccia.
For example, his studies have launched clinical testing of an inhibitor of the hypoxia-related protein known as connective tissue growth factor. In animal studies, the inhibitor stops the spread of pancreatic cancer. Tests in humans begin this summer at Stanford. This is great news, says Giaccia. And if a LOX inhibitor proves successful at stopping the spread of a wide range of tumors, the advantages would be even greater.
“If we could find a way to eradicate primary tumors, we could stop worrying about metastasis,” says Giaccia. “But we’re not there yet.”
An end to metastasis isn’t here yet either. But it’s within reach.

