DNA’s helper is a powerbroker in its own right. Who knew?
Illustration: Tavis
Coburn |
|
 |
|
By AMY ADAMS
Until 1998, geneticists had a frustrating vacancy in their molecular toolbox. They had abundant tools for adding new genes to cells but no simple way to find out what would happen if they took a gene away. That left one side of genetic research more or less unexplored.
Then came a Nature paper by Andrew Fire, PhD, at the Carnegie Institution’s embryology research center in Baltimore, and Craig Mello, PhD, of the University of Massachusetts Medical School in Worcester. This paper showed for the first time that the well-known molecule RNA could slip into a cell, seek out a gene’s “make a protein” message and then destroy it. They called this process RNA interference or RNAi.
Since that paper was published, thousands of researchers have caught the RNAi bug. Fire and Mello had revealed RNAi in their laboratory animal of choice, the nearly microscopic roundworm. But others soon found that it also operates in flies, mice, plants and human cells. So far the only laboratory organism in which RNAi fails to operate is the lowly yeast. In 2002, the journal Science heralded the technology “Breakthrough of the Year.” RNAi research has taken off, particularly at hotbeds at Cold Spring Harbor Laboratory in New York; the Massachusetts Institute of Technology; the University of Massachusetts, Amherst; and now Stanford.
With their new tool in hand, researchers can dream up previously impossible experiments. Eliminate the protein made by a gene and look for cellular changes? Develop gene therapies that quiet disease-causing genes? Knock out all gene products in a cell one by one to look for a particular effect? None of these was possible before Fire and Mello sussed out RNA’s double life. Now they’re practically routine.
Since arriving at Stanford in the fall of 2003 and joining the pathology department, Fire has rallied RNAi researchers from across the medical school and the university, starting a weekly journal club to discuss RNAi research. This meeting might be the only place at Stanford where people studying gene therapy, cell division, viral RNA replication, tumor formation and plant development all enthuse over the same scientific questions. The biggest of those is how best to harness RNA’s gene-disabling identity now that it has been revealed.
Opposites attract
Part of RNAi’s mystery is its very unexpectedness. RNA’s normal role in the cell is to carry a message from a gene to the cytoplasm where it directs a protein-making assembly line. That public life of RNA has been known for decades. In RNA’s covert life, it destroys those very messages and prevents proteins from being made. That’s like finding out your neighbor has a secret life destroying her own landscaping. It caught people off guard.
Illustration: Tavis
Coburn |
|
 |
|
“This changed how people think about doing science,” says Aaron Straight, PhD, assistant professor of biochemistry. He says researchers can now look at the effects of every gene in an organism. “That’s an extraordinary advantage,” he says.
The process of RNAi hinges on RNA’s Velcro-like nature. Like its sibling DNA, RNA is composed of a series of subunits called nucleotides designated A, U, G and C strung together in a chain. The series of letters making up each molecule determines what protein it generates and also allows it to clasp other RNA molecules or DNA. An A on one RNA molecule will find its match with a U on the other molecule and the C’s and G’s pair up. A molecule with the sequence ACUG, for example, would pair up with the opposing sequence UGAC.
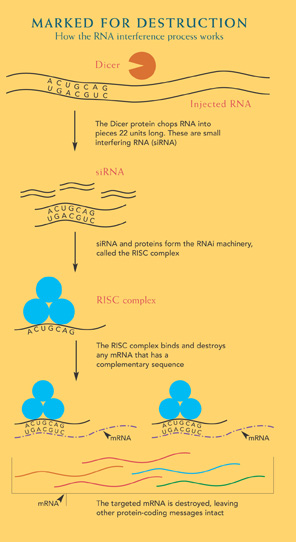
Although the fine details are slightly different in each organism, the broad brushstrokes of RNAi go like this: RNA molecules up to many thousands of letters long enter the cell where a protein called Dicer chops it into units of 21 or 22 letters. These chunks are called small interfering RNAs or siRNA. They conglomerate with proteins in the cell to make up the RNAi machinery. These complexes bind to the protein-producing RNAs within the cell that have a matching series of A’s, U’s, G’s and C’s. This binding marks the RNA molecule for destruction and eliminates the protein.
This knack of completely eliminating a protein makes RNAi a valuable research tool. What was once a laborious and only sometimes successful process is now as easy as sneaking in an RNA molecule with a sequence that matches the RNA you want destroyed. This process works in cells in a lab dish or in whole animals such as worms, where the animals eat the RNA molecules along with their bacterial meal. The process of introducing the RNA in a lab mimics the original role of the RNAi machinery — that of fending off viruses that inject RNA. Dicer and the other proteins chop the virus RNA into short sections that then direct the RNAi machinery to destroy viral protein-coding messages, disarming the viruses.
Silencing disease
Among the early adopters of RNAi was Mark Kay, MD, PhD, professor of pediatrics and of genetics, who realized in 2002 that the technique could greatly expand options for gene therapy. Until RNAi came along, gene therapy was restricted to diseases such as hemophilia or cystic fibrosis where a healthy copy of a gene could replace a mutated gene and cure the disease.
RNAi opened up gene therapy for diseases caused by an overabundance of normal protein. Kay was specifically interested in liver disease caused by infections with hepatitis B and hepatitis C. If he could block the creation of the hepatitis proteins, the virus would be unable to reproduce and the disease-causing population would eventually peter out. Other people are harnessing RNAi to block HIV infections and to stop the overproduction of the protein that drives macular degeneration, the leading cause of blindness. In fact, two companies recently announced that they would start testing an RNAi therapy for macular degeneration in humans.
Kay says his RNAi gene therapy isn’t ready for the clinic, but he has successfully blocked hepatitis B from replicating in mice. “We’ve worked on a gene therapeutic approach against viral hepatitis for about 10 years and this is the first thing we’ve done that really looks promising,” he says.
Unmasking the players
Soon after Kay first put RNAi to work, Straight saw RNAi as a way to speed along his own research. He had been trying to understand the structure of a spot on the chromosome called the kinetochore — a protein complex that latches onto the fibers that pull chromosomes apart during cell division. But finding all the proteins involved proved elusive. With RNAi, Straight could stymie the efforts of each gene in a fly cell one by one. If any lacked a normal kinetochore, Straight would know the muzzled gene played a key role in the structure’s development. With roughly 20,000 genes to systematically shoot down, that process would have been impossible without RNAi.
“I think this approach is great,” Straight says. He and others at Stanford hope to create a dedicated lab to help run and analyze large-scale RNAi experiments such as his. Although he did his work in fly cells, other groups at Stanford have developed ways of doing similar large-scale experiments with cells from frogs and from humans. This ability to shut down each gene in several different organisms could rival the impact on genetics of microarrays — moving research from a one-gene-at-a-time approach to a global view of a cell’s activity.
Illustration: David
Armario |
|
 |
|
Flicking the master switch
Although Fire and Mello worked out RNAi in roundworms, an early hint of RNA’s double life arose during studies of plants. It was also in plant studies where an unexpected second role for RNA, Dicer and the rest of the RNAi machinery came to light — that of turning genes on and off.
In 1990, Richard Jorgensen, PhD, then at DNA Plant Technology in Oakland, was trying to create petunias of a new shade of purple by splicing in an extra copy of the purple pigment gene. But his petunias bloomed white instead. After learning about RNAi years later, Jorgensen and colleagues solved the mystery. RNA from the extra pigment gene had conspired with the RNAi machinery to shut down the purple-producing gene.
“When the petunia stuff came out, those of us who worked in plants thought ‘Wow, petunias are weird; I’m not even going to think about that,’ ” says Kathy Barton, PhD, a staff scientist on Stanford’s campus at the Carnegie Institution’s plant biology department. But she has since gotten her head around the idea of RNA as a genetic workhorse — she has found RNA molecules produced by plants that turn genes on and off as the leaf develops.
It turns out that Barton and the petunia researchers had bumped into short strands called micro RNA, whose sole job is to regulate genes. As with RNA molecules inserted into a cell by meddling researchers, Dicer chops these micro RNAs into roughly 22-letter units. But instead of eradicating a protein-coding message, these small RNAs go to the cell’s nucleus and turn genes on and off. Scientists have now found micro RNAs controlling the fate of genes in humans and in all other animals they’ve investigated.
This role of RNAi in development immediately struck Mike Kaplan, MD, professor of otolaryngology, when he first read of it in 1999. “Cancer is development gone awry, and RNAi is involved in development,” Kaplan says. The department is so intrigued it’s establishing lab space and hiring full-time researchers to begin looking at RNAi misregulation in head and neck tumors.
The potential role of RNAi in disease is a new direction for Fire. As a member of the pathology department, he’s rubbing elbows with plenty of likely disease-focused collaborators, he says.
The first step in this new direction came with the arrival of postdoctoral scholar Weng-Onn Lui, PhD, who is examining whether defects in the RNAi machinery are involved in tumorigenesis. Lui says he’s intrigued by findings hinting that tumor cells hold quantities of micro RNA that vary from levels in healthy cells. Eventually their work could open a new avenue for cancer treatment.
In the end, RNA’s secret life could be its most valuable.
Comments? Contact Stanford Medicine at

